Threads
Observational vaccine data from Scotland
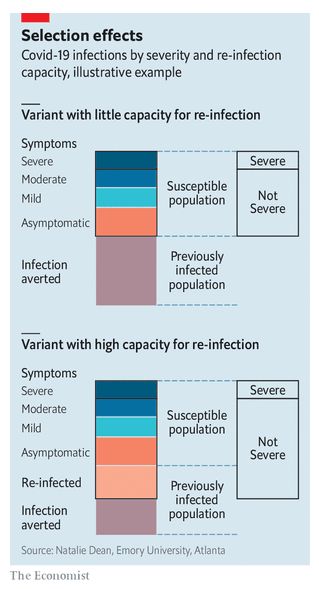
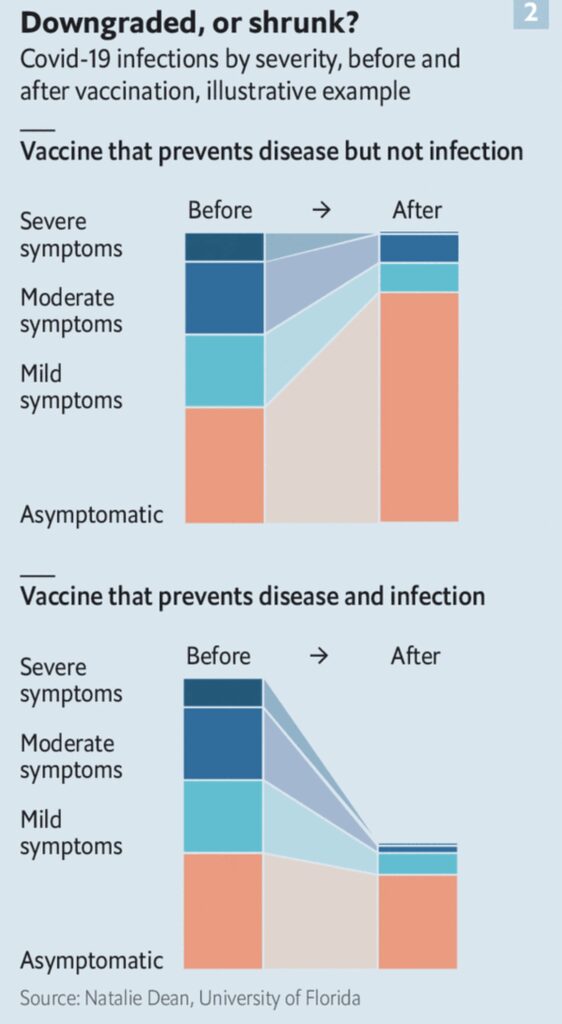
I had the opportunity to write a comment about findings from a large vaccination cohort study in Scotland. I use the comment to discuss some of the challenges of observational vaccine studies and the potential for lingering bias.
— Natalie E. Dean, PhD (@nataliexdean) April 26, 2021
THREAD 1/11 https://t.co/XO39e7Vd3h
Communicating about safety
Regarding vaccine safety, regulators are in a tough position. Transparency is a good principle. But one thing to do better is to limit the dead time between an announcement and the details of its rationale (the media briefing). Here be dragons. 1/3
— Natalie E. Dean, PhD (@nataliexdean) April 13, 2021
J&J pause
A few comments on the J&J news:
— Natalie E. Dean, PhD (@nataliexdean) April 13, 2021
– A pause is not permanent
– The FDA is cautious because it’s important to be responsive to any safety concern; long-term trust is easily lost
– We have additional insights from the AZ vaccine used in Europe, which is also adenovirus-based
1/2 https://t.co/ShdWrBo7rn